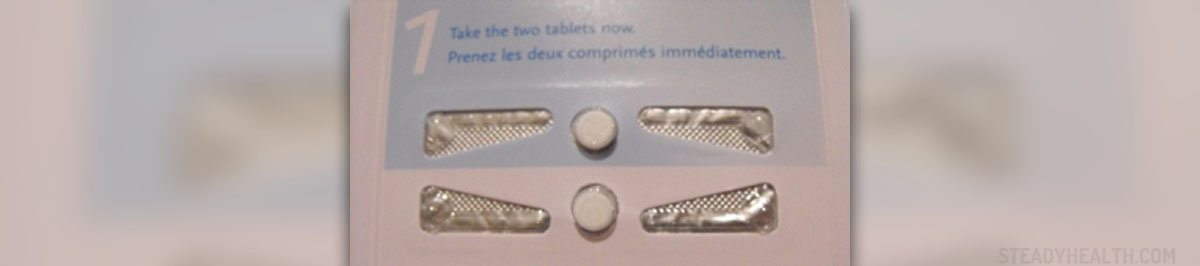
In 2008, the Food and Drug Administration (FDA) already approved the sale of Plan B without prescription to women over 18 along with ID checks that caused controversy. That age was subsequently lowered to 17. Plan B started off as a two-pill emergency pregnancy prevention drug, but has since created a one-pill emergency contraceptive that can be taken up to three days after unprotected intercourse to prevent pregnancy. It's manufacturer tried to widen the pill's access before, but under the Bush administration their efforts (along with those of some women's rights groups) did not result in anything. If approved for over the counter use to girls and women of any age, Plan B could help prevent many unwanted pregnancies, as well as potential abortions.
Conservatives fear that this could lead to sexual promiscuity, but current teenage pregnancy rates in the US clearly show that the "save yourself until marriage" message did not have any effect. The current administration has already taken a different stance look at Obama offers sex education to American teenagers, for instance. Perhaps it is time to recognize that teenagers will have sex, and make sure that those teenagers that need the morning-after pill get timely access to it? It will be interesting to see what the outcome of this will be. Another morning-after pill EllaOne, was already approved for over the counter use last year. The FDA is expected to make their decision about Plan B within 10 months.





,-Or-Ibuprofen-Which-Over-The-Counter-Painkiller-Should-You-Choose_f_280x120.jpg)






Your thoughts on this
Loading...