Fanconi Anemia Prevalence
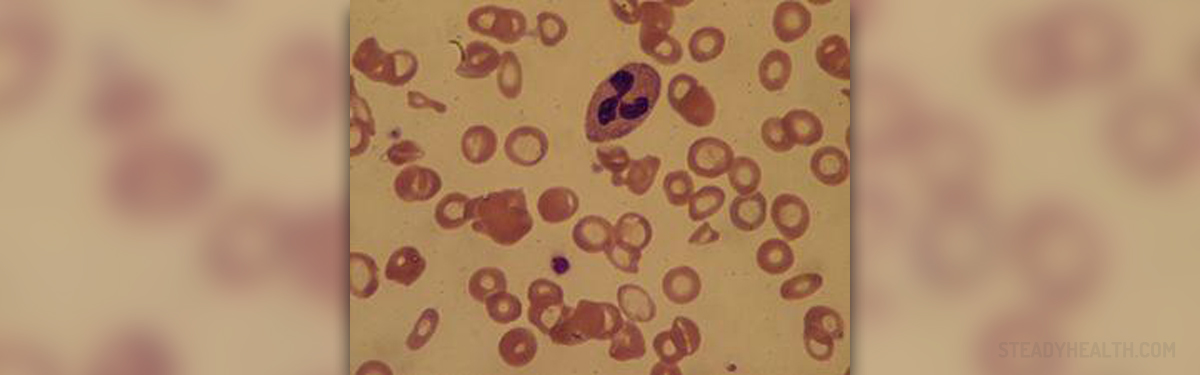
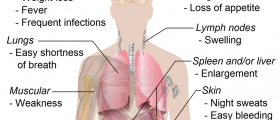
Fanconi anemia is a type of genetic disorder which affects approximately 1 in every 350, 000 newborns, and is especially widespread among the Afrikaners and Ashkenazi Jews. Fanconi anemia was named after a Swiss physician, Guido Fanconi, who first attempted to describe the condition. The genetic mutation associated with fanconi anemia is thought to have first originated in Europe a few millennia ago, and over time was transferred to the American soil. Brazil was one of the places where the defective genes spread quickly and affected a substantial number of people. In addition, when defective proteins that are supposed to repair DNA fail to do so, the resulting condition is fanconi anemia. The two most prevalent health consequences of the disorder are bone marrow failure and leukemia. Most people who suffer from fanconi anemia at some point in their lives will develop bone marrow failure, resulting in the body’s inability to fight off infections, form clots, and transport oxygen. When it comes to the physical features of individuals suffering from the condition, they include abnormally developed arms, eyes, head, ears, and kidneys. Many also suffer from congenital problems, short stature, and endocrine system anomalies. In addition, most individuals with fanconi anemia live up to 30 years of age, or do not reach adulthood. As far as the bone marrow failure is concerned, blood cell growth factors are used as a short term form of treatment but unless there is a suitable donor to prolong the life of the individual the patient cannot live with the condition. As only around 50 to 70 percent of individuals respond to androgens and hematopoietic growth, it is recommended that hematopoietic stem cell transplantation be performed in order to provide a more long term solution. Another relatively often employed type of treatment involves choosing healthy and compatible embryos and taking the blood from the umbilical cord in order to perform stem cell transplants and help affected people. Other than leukemia, fanconi anemia patients are highly likely to suffer from neck, head, gastrointestinal, esophageal, anal, and vulvar tumours. Even if an individual has had a successful bone marrow transplant and is free of the blood related problems, he or she must regularly be checked for the signs of cancer. Other than having to deal with the threat of bone marrow failure and cancer, many fanconi anemia patients are born with physical anomalies, such as missing thumbs. Many also suffer from both physical and mental developmental defects. When it comes to diagnosing fanconi anemia, there have been substantial scientific advances following the disorder. By cloning four out of seven fanconi anemia genes, scientists were able to prove cellular and clinical heterogeneity. Subsequently, molecular diagnosis has become possible for most of the patients, but the diepoxybutane/mitomycin-C stress test remains the most widely used. The way the test works is by comparing the substantial instability of chromosomes observed in FA cells compared to non FA control groups. Also, there have been a lot of research studies concerning fanconi anemia conducted worldwide. In Spain, for instance, clinicians from a university in Barcelona have been studying the disease throughout the country, where about 100 individuals are affected by this rare disorder. The Spanish studies also included patients from Nigeria, Germany, Pakistan, US, UK, Mexico, Brazil, Peru, and Argentina. The researchers concluded that the mutation responsible for the development of the disease in all of the analyzed countries is a form of an Indo-European defect which originated thousands of years ago. The results of the studies also revealed that the cell death and subsequent anemia are caused by a malfunction of a certain FANCA protein responsible for DNA repair and genomic stability. Other than cell problems, the condition also affects tissues, leading them to develop into cancers. Many researchers agree that the genetic study of fanconi anemia is crucial for cancer research as well. The implications of the Spanish studies are vast and include many aspects of the disorder, including diagnosis, prognosis, and future research developments. Many hospitals and universities from Spain and Portugal were actively involved, and so were numerous other European and American research centers and clinics.
Genetic Facts in Fanconi Anemia
Fanconi anemia is an autosomal recessive condition, which in other words means that one defective copy of the gene has to be inherited from the mother and the other defective copy from the father. Also, with each subsequent offspring of the two carriers there is a 25 percent possibility that the child will have fanconi anemia. So far there have been about 15 distinct genes identified which are known to cause fanconi anemia. Most individuals are not aware they carry defective genes so it is always advised that couples who are contemplating having children first go through genetic counseling and testing. It is estimated that about 1, 000 individuals are affected by the disorder worldwide.









Your thoughts on this
Loading...