Many people are too scared or intimidated to go to a doctor to get tested for HIV. In the US, a panel has made a decision that brings us a step closer to the availability of an over the counter, at-home HIV test called the OraQuick In-Home HIV Test.
Experts on the Blood Products Advisory Committee voted unanimously to back the approval of the new at-home HIV test, which will be examined by the Food and Drug Administration later this year. HIV testing is not done by enough people who could be at risk. They said that the test would help HIV positive people gain access to healthcare and social help, and urged the company who came up with the test to place very visible warnings about the possibility of a false negative on the packaging.
Carl Schmid, deputy director of the AIDS Institute, was pleased with the pane's approval of the new test. He told the Associated Press: "We are always looking for game changers, and we believe this is one of them. Not only will it help reduce the number of infections but it will bring more people into care and treatment."
The FDA is under no obligation to follow the recommendations of the panel, but it often does and in 2004 a 99 percent effective version of this test was approved for use by healthcare professionals. The makers of the new at-home test, OraSure from Pennsylvania, said that the test could be sold for around $60. It works by swabbing the outer area of the gums, which apparently is different in make-up to saliva, to test for the HIV virus.
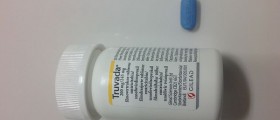
At the same time, another advisory committee of the FDA called the Antiviral Drugs Advisory Committee approved a pill that could decrease the chance of getting HIV for at-risk groups. The pill, called Truvada, is already used by HIV positive people, but could greatly reduce the risk of contracting HIV for people who have an HIV+ partner, the panel said.
Also see: Fertility treatments to prevent spread of HIV safe.
- www.cdc.gov/hiv/testing/hometests.html
- www.nhs.uk/conditions/hiv-and-aids/diagnosis/
- Photo courtesy of Freepik: www.freepik.com/premium-photo/aids-hiv-disease-concept-doctor-wearing-white-coat-blue-rubber-medical-gloves-with-pinned-red-ribbon-as-symbol-aids_6159245.htm#page=1&query=hiv%20test&position=5












Your thoughts on this
Loading...