Center for Disease Control has reported that the side effects of the shingles vaccine include redness, swelling, headache, and soreness.
Shingles Information
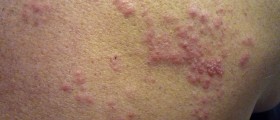
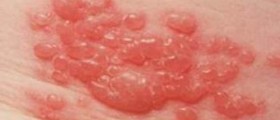
Shingles, or herpes zoster, is a disease caused by infection with the varicella-zoster virus (VZV). The virus that causes shingles is the same that causes chickenpox. Shingles occur when the virus reactivates after years of remaining in a dormant state.Shingles are contagious and may spread from an affected person to those who have not had chickenpox. Initially, shingles manifest with burning pain and sensitive skin that may last for several days. Numbness and itching may also be present sometimes. After about a week, a rash may appear, similar to a chickenpox rash. The rash starts as small blisters filled with fluid and it appears in a band or a small area that may start from the middle of the back around one side of the chest to the breastbone.
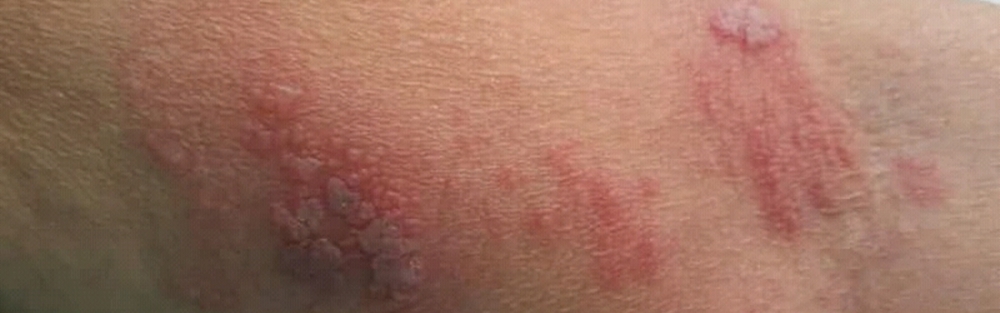
Shingles can cause intensive pain. If early treatment is provided, the severity and duration of the infection can be reduced. Commonly, shingles heal well and rarely cause any complications. However, the incidence of shingles infection and its severity can be reduced with vaccination.
Shingles Vaccine
Shingles vaccine is called Zostavax and it contains live, weakened varicella-zoster virus. FDA in the United States approved the vaccine for people aged 60 and over who have had chickenpox. Scientific studies show that the shingles vaccine can reduce the risk of developing the infection by 50%.
The vaccine also reduces the symptoms and complications associated with shingles. It reduces the risk of postherpetic neuralgia by 67%. It was found that the shingles vaccine is most effective in those aged 60 to 69 years old. The effect of the vaccine lasts at least six years but may be effective even longer.
- There are approximately one million cases of herpes zoster in the U.S. annually, and it is estimated that one in three people will develop shingles in their lifetime. In adults over the age of 85, this ratio increases to one in two. Approximately 1% to 4% of adults who develop shingles may be hospitalized for complications, and the majority of these patients tend to be immunocompromised (i.e., with HIV or cancer, or from immunosuppressant medications).
- One of the most serious complication of shingles is postherpetic neuralgia (PHN). Patients with PHN continue to have long-lasting pain in the areas where the rash appeared, even after it clears up. For some patients, PHN may resolve in a few weeks or a month but for others, it can take years. Approximately 10% to 50% of patients who develop herpes zoster will experience PHN.
- Vaccinating against shingles is the best way to prevent this infection and its complications. The original herpes zoster vaccine, Zostavax® or Zoster Vaccine Live (ZVL), was approved in 2006. However, the efficacy of ZVL in preventing herpes zoster was found to be variable, at about a 70% rate of effectiveness in patients aged 50 to 59 and only 38% in patients over the age of 70. Furthermore, in 2016, only an estimated 33.4% of Americans aged 60 years or older had received ZVL.
- After more than a decade, a new shingles vaccine, Shingrix® (GlaxoSmith Kline) has been added to the market. Herpes zoster subunit vaccine (HZ/su) was approved by the Food and Drug Administration (FDA) in October 2017 for the prevention of herpes zoster in adults aged 50 and older. It is now recommended as the preferred herpes zoster vaccine in immunocompetent adults in this age range by the Advisory Committee on Immunization Practices (ACIP).
- In two phase 3 trials, Hz/su significantly reduced the development of herpes zoster in adults aged 50 and older, and of herpes zoster and PHN in adults aged 70 and older. Because of its superior efficacy over ZVL, HZ/su is now recommended by ACIP as the preferred vaccine for adults aged 50 and older for the prevention of herpes zoster and PHN.
- An additional phase 3 trial showed that Hz/su elicited a strong immune response in patients who were previously vaccinated with ZVL. Therefore, ACIP recommends that all adults 50 and older should receive HZ/su regardless of previous ZVL vaccination.
Side Effects of Shingles Vaccine
A vaccine, just like any medicine entails potential serious adverse effects such as an allergic reaction. However, the shingles vaccine most commonly causes mild side effects. They include redness, soreness, swelling, or itching at the site of the injection. In some cases, a rash similar to chickenpox may develop near the place where the vaccine was injected.
Headache is a less common side effect of the shingles vaccine and is seen in one in 70 vaccinated individuals. According to certain sources, the shingles vaccine may also have side effects like runny nose, fever, chills, diarrhea, and body aches.
Shingles vaccine should not be given to people with weak immune systems due to cancer treatment, immuno-supressing medications, HIV/AIDS disease, organ transplant, leukemia, lymphoma, and other blood or bone cancer. This also applies to those who have had a severe allergic reaction to gelatin or antibiotic neomycin and pregnant women.








Your thoughts on this
Loading...